The Australian research team's latest reconstruction of the evolutionary lineage of reptiles shows that intradermal osteoderms, the small bony plates in lizard skin that look like "chainmail", are not ancient relics as the traditional view suggests, but have arisen repeatedly, independently and even "come back" in multiple lineages over the long history of evolution. This result overturns the consensus in the biological community that has lasted for more than a century.
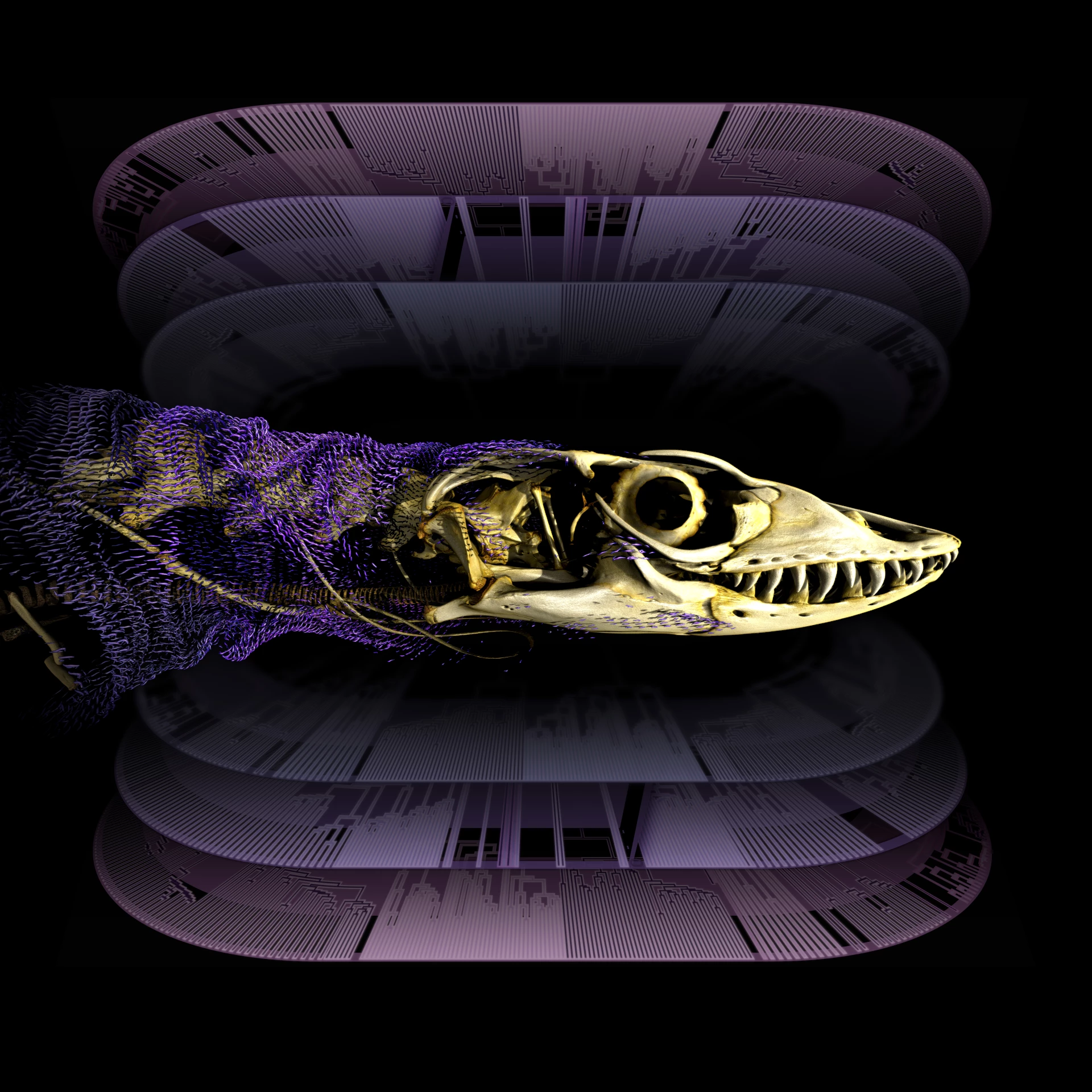
Scientists from the Museums Victoria Research Institute (MVRI) pointed out in the latest research that these bone plates buried in the skin are not ancient features that most lizards inherited from their common ancestor and were subsequently lost in some branches. On the contrary, intradermal osteogenesis in most lizards evolved independently many times in the middle and late stages of different lineages, and some even occurred tens of millions of years after related groups initially lost their bone armor. The research team said that many lizards have "rebuilt" and optimized their protective armor from scratch, demonstrating a rare phenomenon of evolutionary "re-innovation".
This trend is typical of monitor lizards, especially in Australia. Research shows that early monitor lizard ancestors, including the Australian Goanna monitor, completely lost intradermal bone formation during evolution. The reason is probably because the demand for speed and agility in an active hunting lifestyle is higher than the protection provided by thick armor. However, during the Miocene about 17 million to 20 million years ago, some of the common ancestors of Australian and Papua monitor lizards once again evolved lighter and more flexible bony leather armor, which is thought to be related to their spread to arid environments and the face of new ecological pressures. The researchers speculate that in these environments, in addition to providing defense, intradermal osteogenesis may help reduce water loss or strengthen specific parts of the body for behaviors such as climbing.

"This discovery once again highlights Australia's unique position in evolutionary history: not only is it home to dominant marsupials and egg-laying mammals, it is now the only place where the 'return' of intradermal osteogenesis in reptiles is known to have occurred," said Roy Ebel, first author of the paper and a researcher at MVRI and Australian National University.
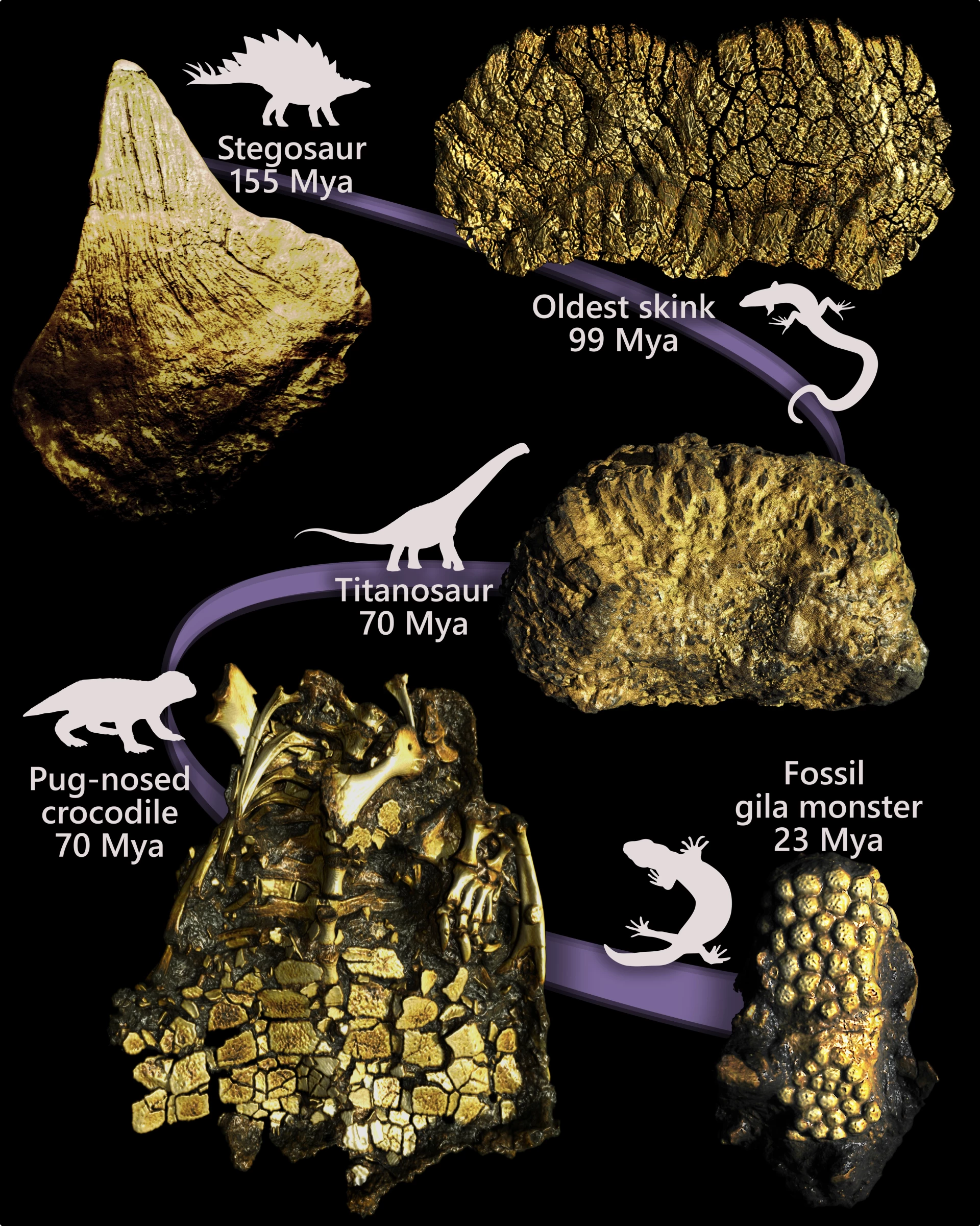
In this work, the research team reconstructed the evolutionary history of intradermal bone formation in squamate reptiles—including lizards and snakes—on a time scale of approximately 320 million years. The scientists comprehensively analyzed 643 living and extinct species, used high-resolution CT imaging data to map the distribution of bone plates on the body surface of different species, and then compared it with large-scale molecular phylogenetic trees and fossil records. They also used a model that allows changes in evolutionary rates in different time periods to deduce whether ancestral taxa had intradermal osteogenesis, thus obtaining the most comprehensive picture of bone armor evolution to date.
The results show that the earliest lizard ancestors almost certainly lacked intradermal bone formation entirely, a feature that remained "absent" for tens of millions of years after lizards first emerged. The study pointed out that intradermal osteogenesis originated independently at least 13 times in various major groups of modern lizards, most of which occurred in a relatively concentrated time window from the Late Jurassic to the Early Cretaceous about 140 million years ago, coinciding with the stage when multiple major lineages began to rapidly differentiate.

Once intradermal osteogenesis occurs in a certain lizard lineage, it is often retained for long periods of time. The study found that in most modern lizard groups, the loss of bone armor in later stages is very rare, which makes intradermal osteogenesis a stable long-term evolutionary feature. Monitor lizards are the only notable exception: they completely lost intradermal bone formation about 72 million years ago, and then reappeared in lighter forms in some Australian and Papuan lineages during the Miocene, forming the "patch" distribution pattern in which both armored and non-armored monitor lizard species exist in the world today.
The study also emphasized that the function of intradermal osteogenesis is far more than just "armor". Unlike bones, these bony plates are formed within the skin and serve multiple physiological and ecological roles. In crocodilians, intradermal osteogenesis has been shown to release calcium when the animals dive for extended periods of time, helping to neutralize accumulated acid in the blood and thereby maintaining acid-base balance in the body. In addition, these bone plates are also involved in processes such as body temperature regulation, mineral storage, and water retention. They are multifunctional biological structures whose evolutionary origins have lacked clear explanations in the past.
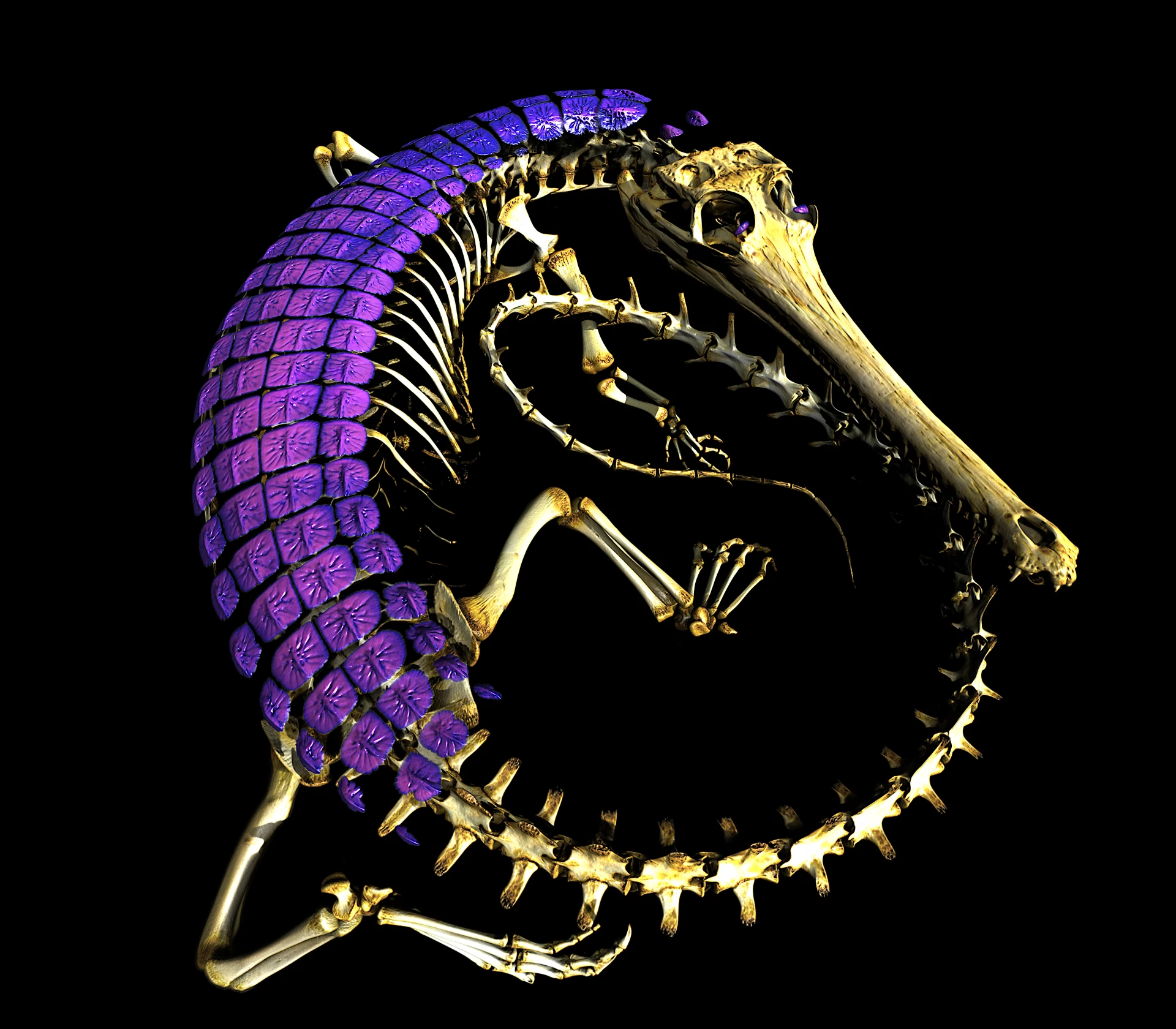
By comparing high-precision CT scan data and phylogenetic modeling of living lizards, the research team was able to draw a timeline of the emergence, disappearance and even reappearance of intradermal osteogenesis in different lineages. Fossil records show that bone armor plates of various shapes existed on the earth as early as more than 100 million years ago. Now this information has been integrated into a unified framework, providing a clearer timeline for the origin and evolution of modern reptile bone armor.
The new results also provide a typical example for understanding the "convergent evolution" of morphology. Research points out that the Gila monster (Heloderma suspectum) native to the United States and Mexico is very similar in appearance to the Tiliqua rugosa (commonly known as the skink) native to Australia. Both are covered with obvious bone armor, but the two have completely different evolutionary paths towards similar body shapes and protective structures. This phenomenon echoes the pattern that flight ability evolved independently at least four times in insects, pterosaurs, birds and bats, suggesting that similar ecological needs can repeatedly "spur" similar morphological solutions in different evolutionary routes.
For zoologists and evolutionary biologists, this new evolutionary map not only answers a century-old debate surrounding the origin of intradermal osteogenesis, but also lays the foundation for subsequent in-depth research on its formation mechanism. Future work is expected to further reveal the genes and developmental pathways that control intradermal osteogenesis, and how these mechanisms are "mobilized" multiple times and independently in different lizard lineages to meet the needs of different environments and lifestyles.
"Our study shows that intradermal osteogenic expression in squamates was acquired multiple times independently," the authors wrote in the conclusion of the paper. "This reconstruction provides the strongest support to date for this hypothesis and also provides a solid foundation for exploring related evolutionary mechanisms." The research team emphasized that these findings help to better understand the selective pressures and evolutionary trajectories that shape the diversity of today's reptiles. Relevant papers have been published in the Biological Journal of the Linnean Society and released through Museums Victoria and academic platforms.