New research shows that specially formulated eye drops can significantly improve near vision difficulties caused by presbyopia (presbyopia). Patients report significant improvements in vision after just one hour of taking the drug, with continued improvements for up to two years, with only mild and temporary discomfort as side effects.
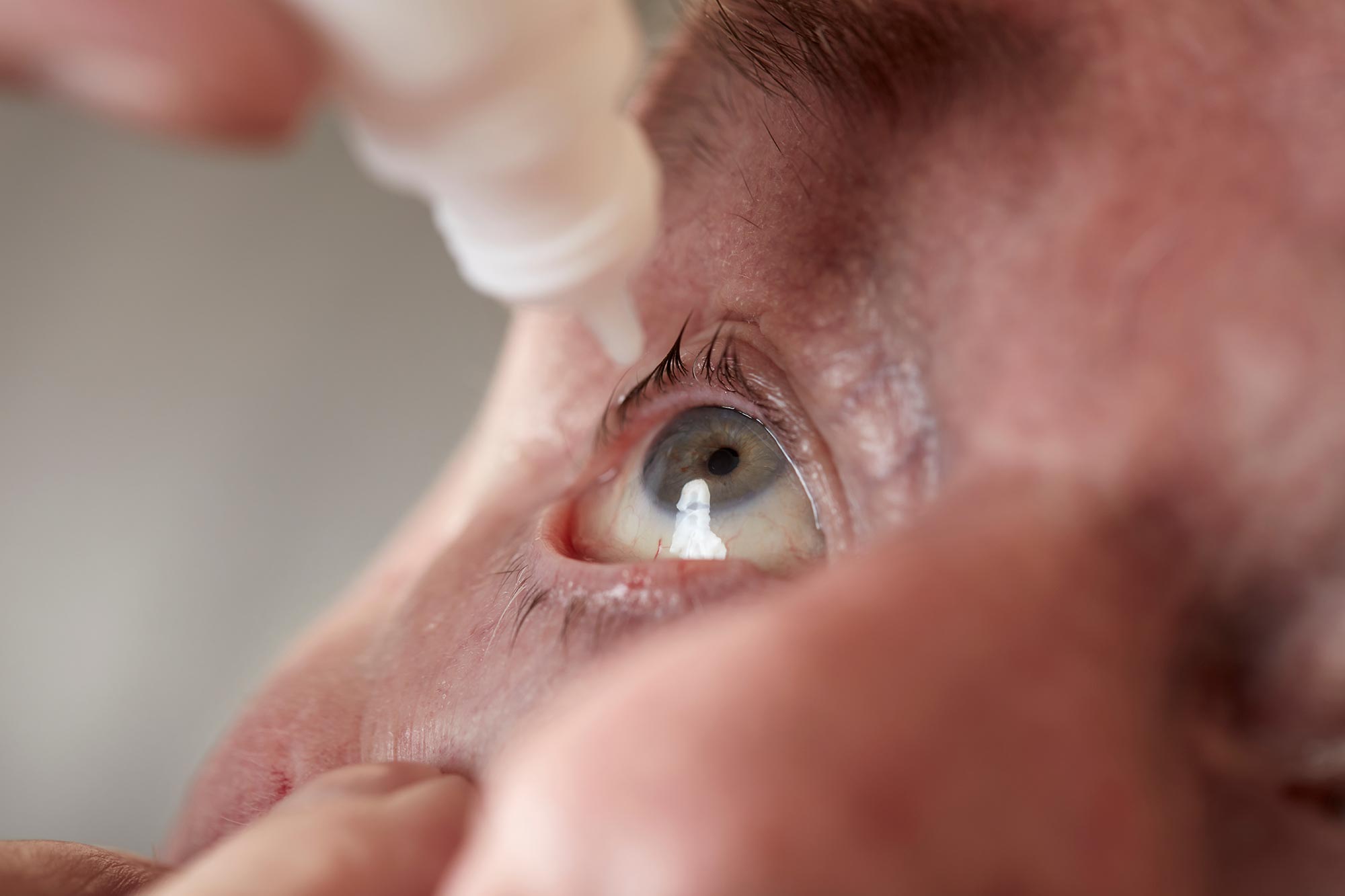
The retrospective study, presented at the 43rd Congress of the European Society of Cataract and Refractive Surgery, included 766 patients with presbyopia. After using the potion, most participants were able to read two, three or more lines more on the Jaeger eye chart for near vision testing, and the effect lasted for two years.
The study was led by Dr. Giovanna Benozzi, director of the Center for Advanced Research on Presbyopia in Buenos Aires, Argentina. She said that in response to the current clinical needs of patients with presbyopia who lack ideal treatment options (such as the inconvenience of using reading glasses, high surgical risks, and some patients are not suitable), the team hopes to prove the efficacy and safety of this innovative drug with clinical evidence and provide patients with a non-invasive, convenient and effective new option.
The eye drop formula used this time was developed by Dr. Benozzi’s father, the late Dr. Jorge Benozzi. The formula contains two active ingredients: the miotic pilocarpine (Pilocarpine), and the anti-inflammatory NSAID Diclofenac (Diclofenac), which reduces the discomfort caused by pilocarpine.
The patient uses the medicine once in the morning and evening every day, and a third time can be added if necessary. A total of 373 women and 393 men were studied, with an average age of 55 years old. They were divided into three groups: 1%, 2%, and 3% according to the concentration of pilocarpine. Diclofenac was used in each group. The evaluation of drug efficacy is mainly based on unaided near vision chart scores (that is, without reading glasses), and is tested one hour after taking the drug and at two-year follow-up.
The results showed that one hour after the first administration of the drug, the near visual acuity of the three groups of patients improved by an average of 3.45 Jaeger levels, and the focusing ability at various distances also improved. In the 1% concentration group, 99% of subjects could read more than two lines; 83% of all patients still maintained good near vision one year later. No significant adverse events, such as increased intraocular pressure or retinal detachment, were observed during the study.
The benefits were obvious in all dose groups: 69% of patients in the 2% group read more than three lines more, and 84% in the 3% group. The median duration of overall efficacy was 434 days. Side effects included temporary dimming of vision (32%), transient irritation during medication (3.7%), and headache (3.8%); no one discontinued treatment due to discomfort. Common side effects of pilocarpine include red eyes, tearing, blurred vision, sensitivity to light, slow focusing, occasional flashes of light or a feeling of floating objects, and in rare cases, retinal detachment may occur.
The study also found that different concentration groups can be individually adjusted according to the degree of presbyopia. Mild patients use 1% concentration for the best effect, while severe patients need 2% or 3% concentration for more obvious improvement.
Dr. Benozzi concluded that this prescription provides a safe, effective, and well-tolerated new option in addition to traditional presbyopia treatment (reading glasses and surgery), which greatly reduces patients' dependence on reading glasses and solves the dilemma of some patients who have neither medicine nor surgery. Of course, eye drops may not completely replace glasses for all patients, but they provide a powerful new solution for patients who want to get rid of the inconvenience of glasses.
The advantages of this study are its large sample size and long follow-up period. It is also the first time that three different concentration combination regimens have been systematically compared. However, since it is a single-center retrospective study, its limitations include generalizability and selection bias.
Burkhard Dick, the elected president of the European Society of Cataract and Refractive Surgery and Professor of Ophthalmology at the University of Bochum in Germany, who was not involved, pointed out that this solution is expected to be helpful to some patients who are not suitable for surgery. However, he also warned that long-term use of pilocarpine may lead to side effects such as decreased night vision, darkening of vision in dark light, and eye fatigue. Long-term use of nonsteroidal anti-inflammatory drugs may also damage the cornea. Therefore, large-scale, multi-center studies are needed to further confirm the safety and efficacy before it can be widely used.
Compiled from /ScitechDaily